The New York Times: Researchers Are Hatching a Low-Cost Coronavirus Vaccine
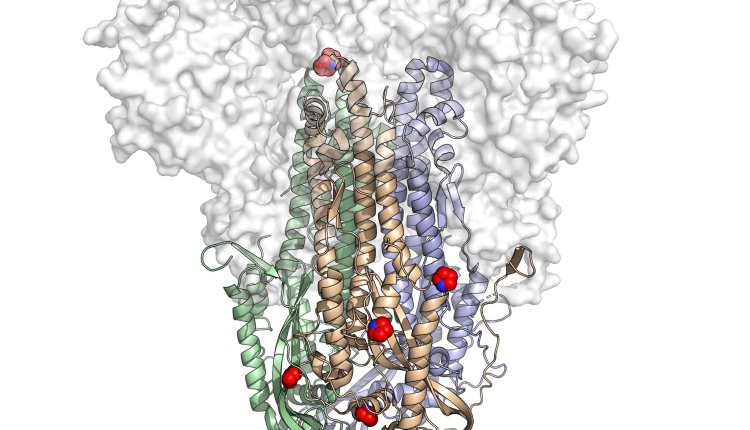
A new vaccine for COVID-19, NDV-HXP-S is entering clinical trials in Brazil, Mexico, Thailand, and Vietnam and could potentially change how the world fights the pandemic. It is widely expected to be easier to make and create more potent antibodies than the current generation of vaccines. The new vaccine can be mass-produced in chicken eggs — the same eggs that produce billions of influenza vaccines every year in factories around the world. If NDV-HXP-S proves safe and effective, flu vaccine manufacturers could potentially produce well over a billion doses of it a year. Low and middle-income countries currently struggling to obtain vaccines from wealthier countries may be able to make NDV-HXP-S for themselves or acquire it at low cost from neighbors. Jason McLellan, a structural biologist then at the Geisel School of Medicine at Dartmouth, and his colleagues were successful in designing a 2P spike unique to SARS-CoV-2. All three of the vaccines that have been authorized so far in the United States — from Johnson & Johnson, Moderna, and Pfizer-BioNTech — use the 2P spike. As Dr. McLellan and his colleagues handed off the 2P spike to vaccine makers, they started looking into ways to improve the vaccine. They came to a decision to implement additional tweaks to the existing vaccine, which resulted in a new spike HexaPro, in honor of its total of six prolines. The structure of HexaPro was found to be even more stable than 2P, more resilient, and better able to withstand heat and damaging chemicals. So far developing countries have received only a fraction of the total distribution of the first-wave vaccine. To that end, the University of Texas set up a licensing arrangement for HexaPro that will allow companies and labs in 80 low- and middle-income countries to use the protein in their vaccines without paying royalties. Read the full article here


Comments are closed.